Turning Leftovers Into Lighting
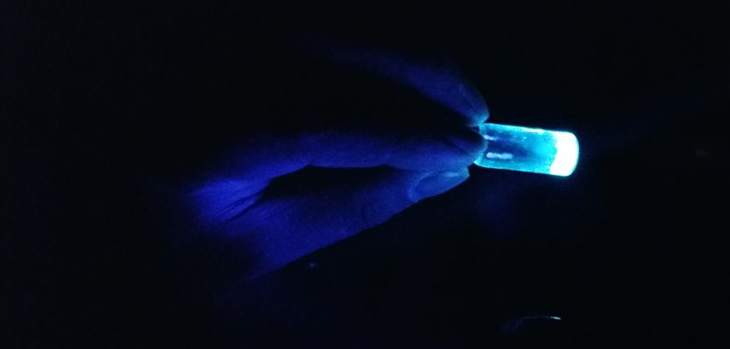
The luminescence of carbon dots can be seen when irradiated with UV light.
Image credits: Prashant Sarswat, The University of Utah
(Inside Science) -- It might be a TV dinner of the most literal kind. Researchers have turned bits of food scraps into light-emitting diodes, or LEDs—those small, glowing devices that illuminate many of today's TV screens.
By heating pieces of tortilla and bread—and even soda and the fumes from burning coal, paper, and wood—the researchers made tiny spheres called carbon quantum dots. These dots glow when you shine ultraviolet light on them, making them useful not only as LEDs, but also in solar cells and as biomarkers, which allows biologists to see specific biological cells light up.
Using food waste could be a cheaper and a more environmentally friendly way to make carbon dot LEDs, said Prashant Sarswat of the University of Utah, in Salt Lake City.
Quantum dots aren't new. In fact, they're being used in some of the latest TVs. They're useful for displays because the dots, much narrower than a human hair, can be tuned to shine in precise colors simply by changing their sizes. Bigger ones are red while smaller ones are blue.
The problem is that they're typically made from semiconductors like cadmium selenide, which is toxic. So in the last few years, researchers have been developing quantum dots using nontoxic and environmentally friendly carbon, which you can find in pretty much any kind of organic material—including leftover food and your unfinished Coke.
Others have used similar heating methods before to turn food and drink into carbon dots. For example, researchers have used waste frying oil, orange and pomelo peels, orange juice, soy milk, coffee grounds, and beer. But most didn't go on and build an LED.
Sarswat and Michael Free, also at the University of Utah, took small chunks of tortilla and bread, soda, and exhaust gases, added a solvent to them, and heated them to temperatures ranging from about 330 to 450 degrees Fahrenheit for 30 to 90 minutes. The heat breaks down the chemical compounds into carbon dots, Sarswat explained. The soda, because it's a simpler substance with pure sugars, turned out to make the best carbon dots.
Making an LED doesn't require too much food or drink.
"It's hopeful that something on the order of a can of Coke or something like that would probably give you enough [carbon dots] for some small LED TV," Free said. The researchers, who recently published their work in the journal Physical Chemistry Chemical Physics, now want to refine their methods so they can better tune the colors of the dots.
"The synthesis process is very cost-effective," said Yogendra Mishra, a materials scientist at the University of Kiel in Germany who wasn't a part of the research. "It can easily be scaled to the industry level." Compared to conventional techniques for making carbon dots, which require pure sources of carbon, using food waste could cut costs by up to a half, he said.
But while these heating methods are relatively simple, the chemistry is not, said Gary Baker, a chemist at the University of Missouri, in Columbia, who also wasn't on the team. Recently, his group used people's urine to make carbon dots (dubbed "pee-dots").
Foods contain all kinds of compounds, and when you heat them, you trigger a smorgasbord of complex chemical reactions and byproducts—in addition to any carbon dots that may form. And those other bits of organic matter also glow. So to isolate the carbon dots and really understand their properties and how they light up, Baker said, you have to purify the solution.
But most studies that turn food into carbon dots skip that step, he said.
"That's a problem with the vast majority of carbon dot papers in the literature."
This new study is no exception.
"If I read this paper as written," he said, "they do absolutely no purification, which is quite rare."
That means you can't attribute all the observed luminescent properties to carbon dots, Baker said.
"We can be very carried away with reporting exciting new results, and we can get ahead of ourselves by studying complicated features of samples that are poorly characterized."
Although the excitement over carbon dots is justified, and this study could be important for future light-emitting devices, the researchers might want to purify their samples and re-analyze them, he said.
"I'm pretty confident it will change their results."

